Rh- Dried Blood Add Solution and It Is Alive Again
Abstract
Courtrooms are asking for reliable scientific evidence in club to forestall wrongful convictions. Thus, a more rigorous arroyo to forensic science canonical by scientific methods is promoted. The written report of human blood dynamics in the context of forensic science is becoming a widespread enquiry topic, although the physics behind wetting and drying of claret is not completely understood. Based on the morphological changes of drying blood pools, the following piece of work presents a patentable method to quantitatively date these blood pools for forensic purposes. As for drying drops of blood, great patterns are observed but they are more disordered. Similar matted crack patterns are observed in the case of gels, their evaporation procedure is, therefore, presented since this topic has been thoroughly investigated. We aim to find reliable patterns that could give information concerning the evolution of a blood pool over time to lead to practical awarding of this knowledge. An empirical model is established betwixt final dried blood patterns and the generating machinery, yielding application in bloodstain pattern analysis for forensic investigations.
Introduction
Bloodstain blueprint analysis is the forensic speciality dedicated to the analysis of blood traces institute on a crime scene1. After a mortality event, a claret pool may be found on the scene. Although some outset suppositions from the ascertainment of claret pools tin can be deduced, the physics concerning the drying processes of claret pools is not yet well known. Some previous studies take focused on the volume decision of a blood puddle for reconstruction purposestwo,iii,4,5 or to make up one's mind if such blood loss could be the cause of death6, only the temporal question of when a blood puddle was formed remains unanswered. The electric current methods used on a crime scene to notice the time at which events occurred are body temperature, rigor mortis, forensic entomology, etc., methods for which the presence of a torso is necessary. If no torso is nowadays, the time estimation of a claret pool formation becomes a crucial slice of information. In other cases, information technology would complement the other available information. Presently, no such method exists. The aim of the present written report is therefore to predict the fourth dimension at which a blood pool is formed with an accuracy of ±xxx mins in gild to add a new slice of data to the global timeline. Furthermore, the evaporation dynamics of volumes of blood greater than a drop take not yet been investigated; therefore, understanding the evaporation mechanisms of blood pools introduces aspects of fluid mechanics of blood which could serve in practical application, bringing a more rigorous approach to forensic estimation7.
Prior work on the evaporation of blood pools examined the morphological changes of blood pools and distinguished five distinct stages8, as shown in Fig. 1:
-
(South.I) coagulation stage
-
(South.Two) gelation phase
-
(S.3) rim desiccation phase
-
(S.IV) centre desiccation stage
-
(S.V) final desiccation stage

Time-lapse of a drying pool of blood from a healthy person, at 23 °C with a relative humidity of 20% showing the drying stages.
It highlighted likewise the fact that a puddle does not dry in a uniform manner, supporting the idea that the shape of the pool is critical in the dynamics of evaporation. To monitor the evolution of drying pools, the report suggested following the drying front, corresponding to the transition border between wet and dry claret, and visually refers to the transition between the reddish and the black colour. This drying front represents an interesting feature to follow with image processing. To sympathize the morphological observation of the drying front end and to develop a reliable tool of interpretation, an understanding of the evaporation mechanism is, therefore, needed.
Evaporation principles
Evaporation corresponds to a mass transfer process of liquid into vapour, which takes identify at the liquid/vapour interface. This process can accept place if the gas phase is not nonetheless saturated, respective to a thermodynamic non-equilibrium. The evaporation of drops with a contact angle of 90° or less have been widely studiednine,10,11,12 and it has been shown that the evaporation depends on the contact line dynamics and is proportional to the radius of the drib. However, for larger stretches of liquids such as a lake, the evaporation is no longer influenced past the triple line. The evaporation tin can exist described equally a steady state authorities where the flux goes from region of high concentration towards region of lower concentration forth a concentration gradient. This is therefore a one dimensional state of affairs, which is described by Fick'south law:
$$J=-D\frac{d\varphi }{dx}$$
(1)
where J is the diffusion flux in mol.m −2.s −1 and corresponds to the corporeality of substance that flows through a square meter during a second, D is the diffusion coefficient in chiliad −ii.due south −1, φ is the concentration in mol.thou −3 and x is the length dimension expressed in thou. In the instance of blood pools, the evaporation appears to be in an intermediate situation between the evaporation of a drop, and ane dimensional evaporation. The height of the pool decreases during evaporation which corresponds to the one dimensional dynamic, but and then desiccation occurs kickoff at the triple line, where the rim dries first. Then this drying front propagates towards the heart of the pool. Really, to describe the shape of a liquid, the capillary length, λ c , is often referred to in fluid mechanics. In general, the social club of magnitude of λ c for most liquids is about a few millimetres. In the case of drops, the radius, which is the characteristic length of this shape, is of the order of λ c , indicating that surface tension prevails over gravity. In the case of larger stretch of liquid (ponds,…) the depth is this time the characteristic length of the shape and is much greater than λ c , indicating that gravity prevails. For the considered pools of this study, an intermediate model has therefore to exist considered, since gravity forces flatten the surface in a pancake shapethirteen, but the edges present a contact bending with the surface. Consequently, to tackle the evaporation problem of a pool, parameters such every bit the thickness of the pool or its shape take to be taken into account. Indeed different shapes volition imply unlike evaporation mechanisms at the triple line. Additional parameters apropos the surroundings accept to be considered likewise during evaporation, such as the surface, the humidity, and the temperature. From a physical signal of view, blood is generally described as a biphasic liquid, with h2o as the fluid matrix and red blood cells (RBCs) as spherical elastic particles of 8 μ m14,15,16,17. Therefore blood is comparable to a colloidal suspension where the interactions between the RBCs are dominated past short range interactions and gravitational forces are negligible. This is comparable to a sol where the interactions between the particles exhibit a Brownian motion. When claret is deposited on a surface, RBCs are evenly distributed and move freely in the aqueous phase. Then blood being ex-vivo, haemostasis occurs rapidly induced by the aggregation of platelets that form a temporary sealant while fibrinogen is being converted to a network of fibrin polymers. Coagulation is known every bit being the innate response to stalk bleeding. It is this response and the formation of the fibrin web that lead to the gelation of the stage. Following gelation, drying takes place with evaporation offset, then improvidence and finally shrinkage.
These dissimilar stages are presented in the Fig. 2. These unlike steps of evaporation remember sol-gel processing with first the formation of a gel, evaporation from the surface followed by evaporation through the porous media. Finally cracking is observed as well. To empathize the evaporation processes of claret, some analogy between a sol-gel transition and blood pools drying could, therefore, exist envisaged.
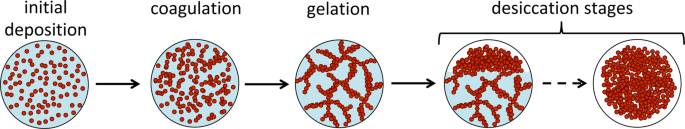
Overview of the different steps of claret evaporation. The filled red circles represent the RBCs while the bluish represents the plasma.
Sol-gel transition and drying
A sol is a colloidal suspension, where the dispersed phase corresponds to solid particles exhibiting Brownian motion. When there is a chemical reaction or during drying, it so evolves into a gel-like diphasic system containing a liquid phase and a solid phase. For the gel-similar backdrop to appear, some of the liquid phase may accept to exist removed by evaporation, depending on the book fraction of the particles present in the sol. Since the literature concerning claret pools drying is deficient, but many studies depict the drying of gels18, we reviewed the literature of sol-gel transitions in society to afterwards compare it with drying blood pools and see if some similarities exist.
One of the first observations concerning the sol-gel transition is that the process goes through different stages. Start the sol transforms into a gel, which presents gummy and elastic properties. The clusters grown either by polymeric condensation or by particle aggregation assemble together to class a giant cluster called gel. Once the gel has formed, evaporation then takes identify through this porous media. Outset the body of the organisation shrinks and the book loss corresponds to the volume that has evaporated. After a critical bespeak the body becomes rigid and stops shrinking. The liquid then recedes inside leaving almost the surface air-filled pores. All the same evaporation continues to occur at the surface. Finally when only a small-scale amount of liquid is left in some isolated pores, evaporation can merely take place inside the system followed by vapour improvidence to the outside. Consequently drying of gels can be divided into several drying stages. Already in 1986 Dwivedi examined this process in some particular by studying the drying process of alumina gels of different thicknesses focusing on the mass loss over fourth dimension. As a outcome he plant that near vii% of the initial mass of gel is left after drying. In the case of blood pools, information technology was similarly shown that 23% of the initial mass is left after drying8. Dwivedi then looked at the dissimilar drying stages and institute that alumina gels undergo three drying stages. The first stage is a constant rate menstruation (CRP)19,xx,21 where the volume subtract of the gel is equal to the volume of liquid lost by evaporation. In this stage, the evaporation rate appears to be comparable to that of an open dish of water22. A critical point is reached at the end of this stage, inducing the shrinkage to stop. Subsequently to this, cracking of the gel is more than likely to occur. After the critical point, gels undergo a offset falling rate (FRP1) where the liquid flows through partially empty pores, followed past a 2d falling rate (FRP2) respective to the final stage of drying. In this final phase, evaporation occurs inside the body and the liquid diffuses to the surface in the course of vapour. The clarification of the sol-gel transition and the drying of inorganic gels represent a classic work where the mechanisms are well established19. Therefore, examining its characteristics is interesting since we tin can envisage that the drying process of blood is likely to present some similarities, which will now be presented.
Materials and methods
Claret properties
A certified nurse sampled claret in 9 ml dried evacuated collection tubes. Blood collection was carried out and approved in accordance with relevant guidelines and regulations of the university and of the National Scientific Enquiry Council. Informed consents were obtained from all volunteers. The tubes are neutral tubes, without any anticoagulant or activator, and pools are created within 30 seconds later on sampling in order to prevent anticoagulant agents interacting with the action of fibrinogen and clotting and to recreate existent bleeding events. The blood is slowly deposited from the tube onto the surface, a few millimetres above the surface. An additional blood tube was sampled for heamatological analysis, but in a coated tube with an anticoagulant. Using heamatological assay (Mindray, BC 3600), the heamatocrit value is determined. For women the heamatocrit level was between 36.2 ± 0.1% and 42.6 ± 0.1%, and for men between xl.1 ± 0.1% and 47.1 ± 0.1%.
Experimental set up-upwards
In social club to accurately follow the drying of a blood pool, the environment must be controlled and kept as constant equally possible. Thus, blood pools were created in a glovebox (Jacomex T-Box, V = 350 L) where the humidity can exist controlled. The experimental room was regulated by air conditioning, allowing the temperature to remain abiding. 3 different surfaces were used, a linoleum surface, a varnished wooden flooring surface and a tile. The chosen surfaces are non-porous surfaces ordinarily plant in criminal offense scenes. The initial mass of the pools varied betwixt 0.30 ± 0.01 g to 31.37 ± 0.01 g. To mensurate the initial masses, and the mass loss during drying, the surfaces were placed on a residuum (Mettler Toledo, MS6002TS). For the offset fix of experiments simply one residual and one surface were used, but for the second set, another surface was placed on some other balance (Mettler Toledo, MS6002TS) next to the first i. The get-go prepare of experiments was to identify the different phases of the drying procedure. The aim of the 2d set was to compare two pools drying in the aforementioned conditions of either same weight but with different shapes or of aforementioned shape but different weights. A photographic camera (EOS 7D digital photographic camera, resolution: 5184 × 3456 pixels) was suspended above the pools and pictures were recorded every two minutes. A surface reference length was placed side by side to the pools in order to make up one's mind later their surface areas.
Results and discussion
Thermodynamic aspects of drying
Drying implies ii types of transfer: oestrus transfer from the surroundings towards the liquid and a mass transfer from the system towards its surface then into the surrounding air. Blood temperature inside the trunk varies from 36 °C to 37.5 °C. To observe the temperature changes of blood when taken from the body, two thermocouple probes were placed, one in the blood pool, at the center of the pool, and another i simply to a higher place the blood pool. The temperature in the glove box was kept constant at 27.two °C. In one case blood was deposited on the surface, at that place is starting time a very fast increment of the temperature measured by the probe. It reaches 31 °C. This corresponds to the transient heat transfer of the warm blood touching the surface at room temperature. The temperature decreases then rapidly and reaches a plateau of 26.iv °C. It takes about 25 mins for the claret to reach this plateau value, which could be attributed to the heat diffusion/convection inside the pool. Then the observed plateau appears to be approximatively ane °C lower than the temperature measured just above the pool itself equally shown in Fig. 3. This is in agreement with blood evaporation as an endothermic procedure. This temperature difference is stable over time, until the pool starts cracking. One time the cracks reach the probe placed in the puddle, the probe is no longer in the liquid part of blood, and the measured temperature goes support to the ambient temperature. This temperature is the same 1 as the one measured with the other thermocouple placed just higher up the puddle.
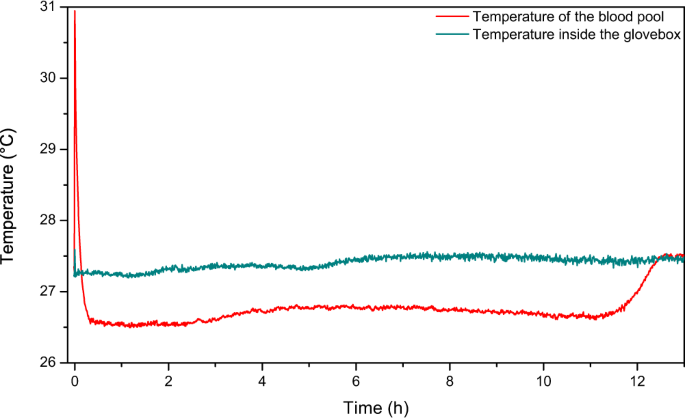
Recorded temperature of a drying claret puddle within the glovebox and of the temperature just above this aforementioned puddle; thousand i = viii.75 g, HCT = 46.7, and 20% humidity.
Evaporation dynamics
Claret is comparable to a colloidal solution where plasma corresponds to the aqueous phase since plasma is a h2o based solution, composed of ninety to 92% water, of vii to viii% plasma proteins and the last 1 to two% are trace amounts of other constituents23. Therefore a showtime unproblematic study was made to follow the mass loss of the blood pools over time and to compare them to pools of h2o. Natural water evaporation is of course a well known phenomenon, just to our best knowledge actual research papers accept focused mainly on natural evaporation from open water, ponds or water baths but no work has focused on the drying of a water pool formed by natural spreading. To compare water evaporation to blood evaporation, blood and water pools were created in parallel and dried in exactly the same weather. Two dissimilar surfaces, linoleum and varnished oak wood, were tested in two dissimilar humidities of xx% and 30%. Since the pools had different drying durations, t f , and initial masses, one thousand i , the obtained results were normalised equally presented in Fig. iv in order to compare both types of pools.
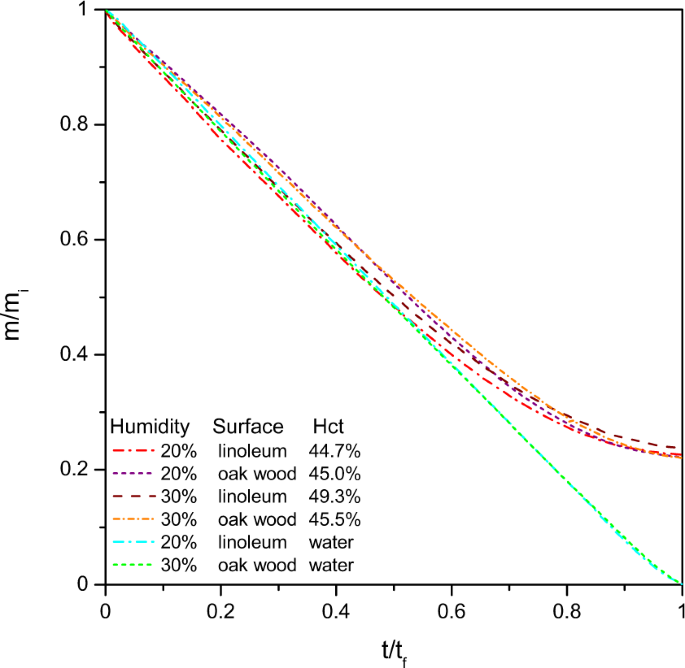
Normalised mass as a role of normalised time of the blood and h2o pools. Experiments were carried out at 20 or 30% humidity, on linoleum or oak wood surfaces. The heamatocrit values of the blood ranged between 44.vii and 49.iii%.
The initial mass loss for water or blood pools is linear until it reaches approximatively 60% of mass loss. At this stage, the pool is pinned to the surface and the liquid phase evaporates at the surface. The volume loss corresponds then to a decrease in acme. When for water the mass loss keeps diminishing in a linear style until 0% of mass is left, for blood pools the mass flow rate decreases significantly until the mass left corresponds to approximatively 23% of the initial pool mass. This amount corresponds to the biological deposit, explaining the small variations that are observed according to the initial heamtocrit value. The same percent was already observed in the instance of claret drops24.
To investigate further the mass loss over time of the pools, evaporation rates were experimentally analysed. To observe an evaporation charge per unit, the mass loss of a pool, which corresponds to a h2o mass loss, is evaluated over time per unit area. The pools were weighed at half-dozen to 12 min intervals during the initial drying stages and xx to 30 min intervals at later drying stages. The evaporation rate corresponds to the recorded mass loss over the time interval per unit area of the total pool area. The evaporation rate is named J* since it corresponds to a mass diffusion flux. Figure 5 presents the obtained h2o evaporation charge per unit of a blood pool of initial mass m i = 31.37 grand as a part of the per centum of water left in the claret. The puddle was dried at a temperature of 23.ix °C and at a humidity of 20%.
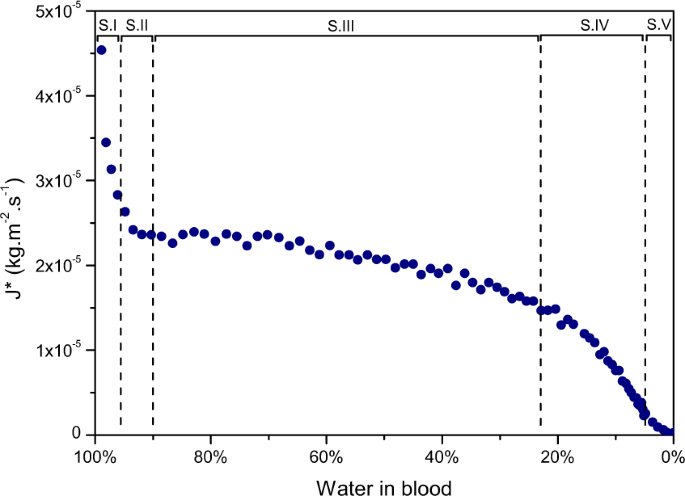
Rate of water loss against the water contained in the pool for a blood pool; m i = 31.37 chiliad, HCT = 42.one%, T = 23.nine °C and 20% humidity.
By looking at the evaporation rate of the blood puddle in Fig. five every bit a function of water left, the evaporation can be compared to the dynamics of alumina gels, where the evaporation charge per unit equally a part of percentage of water left was examined22. Some similarities are clearly noticeable. Just afterwards the pool formation, the rate of water loss decreases fast until coagulation of the phase S.I and and then reaches a abiding rate until the end of stage Southward.III. This recalls the first drying phase of gels, CRP. Then in that location are successively two charge per unit decreases too, S.IV and S.V, similar to the falling rate periods FRP1 and FRP2, until all the h2o present has fully evaporated. During these last stages, evaporation no longer takes identify at the surface but within the porous media. The original liquid is so transported in the form of vapour to the outside, which explains the decrease in the evaporation rate. In the final phase, cracks start being observed. The crack patterns of an unabridged blood pool are quite different from a single blood drop, where the cracks are elongated and directed towards the centre of the stain. Still these cracks show similarities to the ones observed by Pauchard et al.25 during the drying of a colloidal interruption having an ionic force of I = 0.four mol.50−1 or the drying of latex particles 0.1 μchiliad in suspension26. These cracks follow the drying forepart and the separation between them depends on the thickness of the sample, the physical and chemical backdrop, the adhesion with the surface and on the desiccation atmospheric condition26,27. Although the main mechanisms and evaporation of a blood pool seem to exist similar to that of a gel, the characteristics of a puddle have to be analysed as well since in practical cases the shape and size could vary significantly.
Shape influence on the evaporation rate
In order to study the influence of the shape of a puddle on its drying, Twenty pools of the same initial mass, five.24 ± 0.47 g, but of different shapes were created. These were dried in the same humidity of xx%, at the same temperature of 23 ± 1 °C and on the aforementioned surface, a white tile. The initial mass, 1000 i of a pool is known, thus the book, Five, can exist calculated using the blood density:
$$5=\frac{{m}_{i}}{\rho }$$
(2)
The surface area, A, and the perimeter, P, of each puddle were measured using the imageJ software. From this, the summit, h, of the puddle can exist estimated with the following approximation:
These dimensions characterise a blood pool. An piece of cake dimensionless factor can therefore exist deduced, which correlates these shape backdrop. This shape factor named L* is but divers according to the following equation:
$${L}^{\ast }=\frac{A}{hP}$$
(iv)
with A, the area, h, the height and P, the perimeter of the puddle. The shape gene L* was calculated for the twenty pools. An boilerplate evaporation rate for each pool was besides estimated after l% of the initial mass had evaporated. This was in order not to have results biased by the final stage of evaporation, which is very slow and could induce some errors in the overall rate interpretation. The obtained evaporation rates were different for each pool despite the fact that the pools had a like book, and were drying in similar conditions. To take into account the shape factor, the evaporation rates (in kg.m −2.s −i) of the pools were then divided past the shape factor Fifty*. Every bit a result a linear variation of the modified evaporation rates equally a function of the contact angle of the pool was observed (Fig. half-dozen).
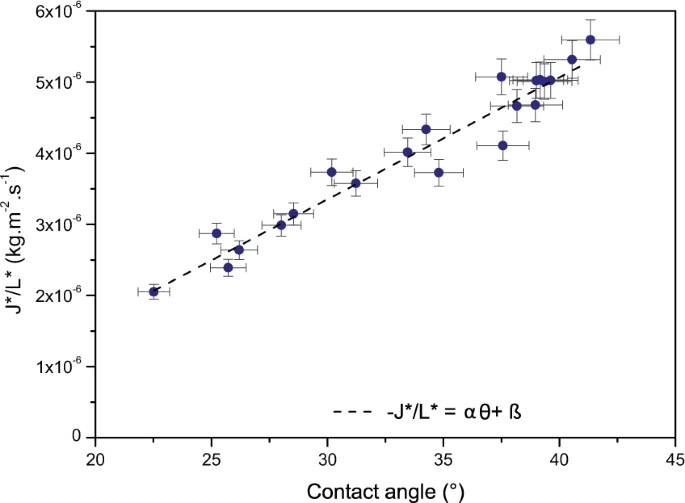
Evaporation rates for blood pools of initial masses five.24 ± 0.47 g divided by their corresponding shape factors L* as a office of the contact bending of the pools. The pools were dried at xx% humidity, T = 23 ± ane °C, on the same surface, a tile. The dashed line corresponds to the linear relationship.
The linear variation is described by: J*/L* =αθ + β, with α = 1.72.10−seven and β = 1.79.10−6. To calculate the contact angle of the puddle the following approximation8 was used:
$$\theta =\arccos \left(\frac{g{m}_{i}^{2}}{2\gamma \rho {A}_{i}^{ii}}+1\right)$$
(5)
The obtained results in Fig. 6 support the idea that the shape of the pool has an influence on the drying dynamics and tin be expressed in the class of a linear relationship. This linear relationship is valid for pools of similar weight, drying at same humidity and temperature.
Size influence on the evaporation rate
To study the influence of the size of a pool on its evaporation rate, we compared pools of different initial masses drying in similar conditions, i.e. at the aforementioned humidity of xx% and the similar temperatures of 23 ± 1 °C on the same surface, a white tile. In Fig. seven, five pools have been selected, whose characteristics are given.
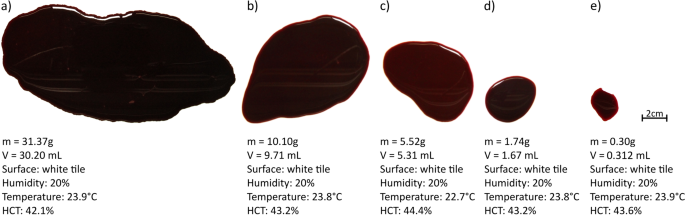
Characteristics of the five different pools used to illustrate the study on the influence of the size.
The rate of water loss equally a function of water left in blood for the 5 pools is compared in Fig. 8a, and in a normalised way in Fig. 8b. The normalised results show obvious different dynamics for the pools having a very depression m i compare to the other pools exhibiting very similar evaporation dynamics. The smallest pool of 0.30 grand approaches the size of a large drop. It presents the highest evaporation rate. Moreover the dynamics present first a plateau evaporation rate until a sharp decrease in the evaporation rate is observed at the very end. The evaporation dynamics of deposited blood drops are said to exist dependent on the contact bending, the vapour concentration and the radius of the drop. The total drying time of a deposited drop of blood beingness relatively short (of the order of half an hour) the observed concluding pattern is the results of a time-scale contest between the thermo-capillary convection and the movement of the particles that takes places in evaporating drops of blood. Some studies have emphasised the role played by Marangoni period to keep the evaporation flux abiding in the kickoff evaporation phase28. After deposition, RBCs are homogeneously dispersed inside the drop. The radial flow triggers the redistribution of RBCs towards the periphery of drops leading to the biological deposit24. Afterward a critical evaporation indicate and clotting (occurring from 2 to 8 min after deposition), Marangoni menses is no longer possible inside the drop and RBCs are trapped into the primal area of the driblet. Equally a event, the evaporation rates decreases significantly until consummate evaporation of the drop. This model appears to no longer be valid for larger amounts of blood, as tin be seen on Fig. 8b where the evaporation stages exhibit different dynamics for the other pools. The evaporation rate of water loss appears to decrease every bit the mass of the pools increases (Fig. 8a). For smaller blood volumes, evaporation is faster. This could be explained past the humidity straight surrounding the pool coming from the modify of state of the aqueous phase into vapour and which is less meaning for small blood volumes. Inversely, the greater the size of the pool is, the more difficult information technology becomes for the aqueous phase to change into vapour since the surface is already saturated with vapour. However, Fig. 8b shows that all pools undergo the same evaporation stages, from the small-scale pool of m i of 1.74 g to the big puddle of m i 31.37 m.
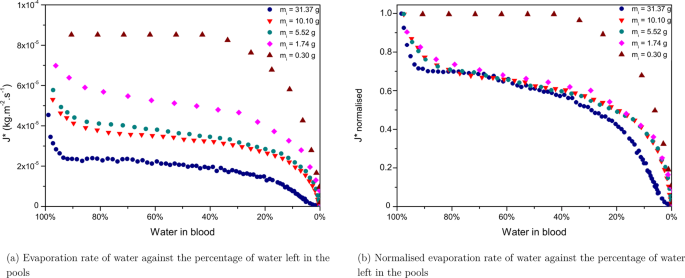
(a) Evaporation rates and (b) Normalised evaporation rates of pools having various initial masses (one thousand i ), drying at 20% humidity, T = 23 ± 1 °C, on the same surface, a tile.
The surface area considered for the evaporation charge per unit corresponds to the full expanse of the pool, but when the pool starts desiccating the wet surface area decreases, which explains the boring decrease of the evaporation rates towards the cease of evaporation, when most 25% of water is left in claret. This resembles the dynamics observed in the case of gels suggesting that with increasing claret book, at that place is a shine transition betwixt drop-like evaporation dynamics to gel-like evaporation dynamics. The fact that the final falling rate is mainly visible for the largest puddle strengthens the assumption that pools of greater volumes tend towards gel-similar evaporation dynamics. This graph illustrates very well how increasing the book of a pool affects its evaporation rate.
Overcoming size and shape influences
In the previous section the evaporation rate of water was calculated using the flow rate per unit area, using the total expanse of the pool. To overcome the influence of the size of the pool, it could exist envisaged to calculate the evaporation using this time the moisture area. Past using the photographic analysis that were recorded during the experiment, the area of the entire puddle and of the wet area of the pool could be measured as a role of time as illustrated with one pool in Fig. ix. In this effigy the entire area of the puddle remains constant with time except later on 25 h respective to shrinkage of the pool when cracking occurs. The wet area, delimitated by the drying forepart, is abiding at the showtime and and then decreases in an about linear style.
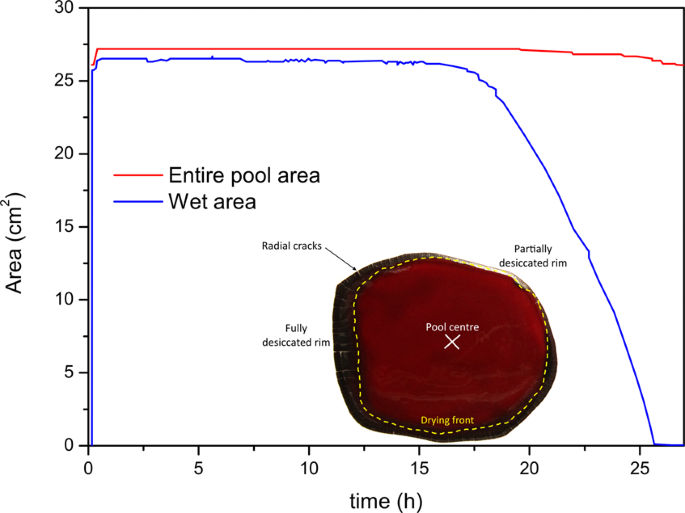
In ruddy: area as a function of time of the entire pool; in blueish: moisture area of the pool as a office of fourth dimension; grand i = five.41 g, HCT = 40.0%, T = 22 °C and 60% humidity.
To summate the evaporation rates using the wet areas instead of the total areas, the measurements of the wet areas of the 4 pools (a), (b), (c) and (d) were used. Pool (eastward), was excluded for this part of the study since its evaporation dynamics were closer to the one observed for drops. The obtained evaporation rates are presented in Fig. x. The evaporation rates of water appear now to be constant. Although, at the very starting time and at the end of the process, the values no longer follow this plateau. Before blood coagulation, the evaporation dynamics are likely to arroyo the dynamics of water, whereas afterwards coagulation those dynamics should be closer to the dynamics of a gel, which explains that there'due south a small decrease at the beginning until reaching a plateau. Then, the evaluation of the evaporation rates is biased towards the finish since the image analysis software can not notwithstanding determine very precisely the wet area when it comes to the final phase of drying.
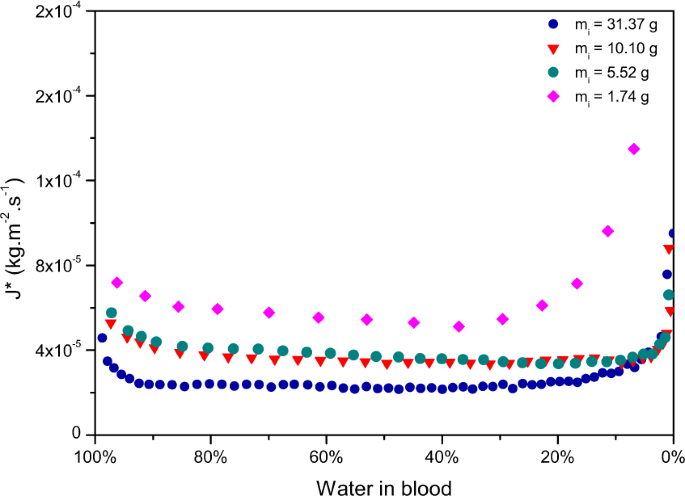
Evaporation charge per unit of water against the percent of water left in pools of various initial masses (g i ), drying at 20% humidity, T = 23 ± 1 °C, on the aforementioned surface, a tile.
This abiding value of the evaporation charge per unit of water every bit a part of water left in blood implies that using the expanse analysis the dynamics can be predicted. Yet the plateau appears to be dissimilar for the four pools. Several parameters must act. A unproblematic relation describing the evaporation rate of a free surface based on convection already exists, given by29:
$${J}^{\ast }={K}_{i}\frac{MPw}{RT}$$
(half-dozen)
with 1000 i the transfer coefficient of the liquid state into the gaseous state (in k.s −1), M, the molar mass (in kg.mol −i), P due west , the saturation vapour force per unit area of water at the surface (in J.m −3), R the universal gas constant (in J.mol −1.Chiliad−1), and T, the temperature (in K). This equation shows that in the case of evaporation of a complimentary surface, the evaporation charge per unit is dependent on the saturation vapour pressure and on the humidity. This equation can be rewritten equally:
$${J}^{\ast }\frac{RT}{M{P}_{due west}}={M}_{i}$$
(7)
The obtained value of the transfer coefficient K i is in thou.s −1. In order to approach the evaporation of blood pools with Fick's Law, the diffusion must be described. Therefore a diffusion coefficient (in m 2.s −one) should be analysed instead of a transfer coefficient (in chiliad.s −1). And so, to obtain a diffusion coefficient of the pools, the transfer coefficient was multiplied past a feature length. The Knudsen layer, L K , was used equally feature length since it corresponds to the very thin evaporation layer of vapour near the liquid, which is described by the post-obit equation:
$${L}_{k}=\frac{kT}{\pi {d}^{2}{P}_{a}}$$
(eight)
where k is Boltzmann'south constant, T, is the temperature, d is the molecular diameter and P a is the atmospheric pressure. Therefore to obtain the diffusion coefficient of each pool, Grand i should be multiplied past the corresponding Fifty 1000 . In the case of blood pools, evaporation depends on the shape of the pool, since the evaporation rate is modified compared to an infinite stretch of fluid. To compare the calculated diffusion coefficient of the pools, the shape gene should be taken into business relationship. As a results the evaporation rate is weighted by the universal gas abiding R, the temperature T, the molar mass, M, the saturation vapour pressure, P west , the Knudsen layer, L K , and in fact by the square root of the shape gene L*. Every bit a effect, the plateaus observed in Fig. 10 scale together to a unique plateau of approximately constant value of 1.10−9m 2.s −1 equally shown in Fig. 11.

Evaporation rate of water weighted by RTL k L*1/2/M P w against the percentage of water left in pools having diffirent initial masses (yard i ), drying at 20% humidity, T = 23 ± 1 °C, on the same surface, a tile.
This plateau could bring an approximation of the diffusion coefficient of blood pools. Information technology appears to exist lower than the ane measured for a driblet of blood30 of 9.8.x−8 thou 2.s −1 but in good agreement with the values obtained for improvidence of colloidal gel of stacked particles into air31, where values ranged between ten−nine and x−10 m ii.s −1.
The various characteristics of a puddle added to the variability of the surrounding parameters pb thus to a very complex problem. Information technology appears that all these parameters are related together, every bit would suggest the results presented in Fig. 11. This last result presents a very interesting approximation suggesting that drying blood pools are very similar to gels. Although the pools were stale in like weather condition, mainly same humidity and same temperature, these parameters should be considered in further studies, as they are known to accept an influence on evaporation. With temperature increase, diffusion coefficients are known to increase. This suggests that temperature can impact profoundly the evaporation rates. Relative humidity affects evaporation as well since diffusion coefficients are known to decrease as the relative humidity increases. Further research should be devoted to seek out on how could temperature affect the plateau obtained for pools drying at 23 ± 1 °C. Fundamentally information technology tin can exist accepted that the shape of a pool and the surroundings parameters are key to understand the evaporation rates. Undeniably the geometry affects the evaporation rate since some geometry would favour evaporation. For 2 pools of identical volume, the round pool will take its Knudsen layer saturated in vapour faster than the thin and stretched puddle. The evaporation of the thin pool will be favoured, it will thus take a greater evaporation rate. Although estimating the geometry of a pool exhibits some doubt, the fact of taking the geometry in consideration already erases a large amount of errors. Moreover the obtained correlation tin can find very interesting practical application.
Forensic estimation
The observations made during this study highlight the complexity of the problem of drying blood pools. Although many questions remain, some interesting features accept been characterised, peculiarly the drying front end. This detail feature presents an interest to BPA analysts since information technology can hands be monitored by photography. In practice, investigation teams demand tools, which tin easily exist brought on the scene, and that will not induce an alteration of the evidence. Photography is a tool beingness already used. The aim of this piece of work was thus to develop a method that would require only photographies so that it could be concretely applicable. Using this applied field tool, and the previously obtained constant value of the improvidence coefficient for drying blood pools, the time at which a puddle was formed tin can be calculated for a pool drying in the same conditions. The puddle presented in Fig. 12 was dried at 22.5 °C on a white tile. The drying front is already clearly visible in this photograph.

Pool drying at 22.5 °C on a white tile.
First, the evaporation rate J * of this pool can exist calculated using the approximate abiding value D blood = i.x−9m two.s −ane, corresponding to the plateau observed in Fig. 11.
$${J}^{\ast }={D}_{claret}\frac{K{P}_{due west}}{{Fifty}_{k}RT{L}^{\ast \mathrm{one/2}}}$$
(9)
50 * is obtained with
$${L}^{\ast }={A}_{i}/hP$$
(x)
where A i is the full area of the pool, and P its perimeter. Both tin can be measured in the picture show of Fig. 12. h is estimated from the average value of the elevation measured for this surface for thirty different pools and yields a value of one.44 ± 0.xix mm. Next, to correlate the calculated evaporation charge per unit with the elapsed time, δt, since this pool was formed, the mass variation is needed. m i can be obtained with
$${m}_{i}=h{A}_{i}\rho $$
(eleven)
Then t 10 corresponds to the fourth dimension at which the motion-picture show was taken after puddle formation such that δ t =t x − t i . m x is the mass of the pool at t ten . Since the wet area, A 10 , at t x tin exist measured, a relation between area and mass is necessary. The following graph in Fig. 13 presents the normalised area equally a part of the normalised mass for the pools studied in the previous department.
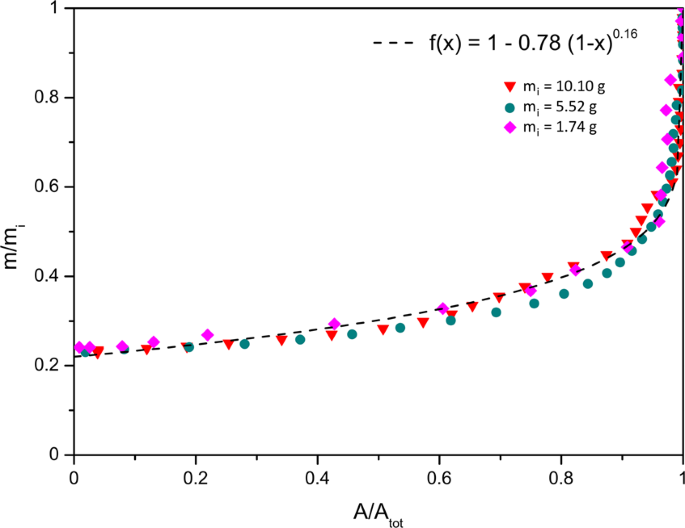
Normalised mass every bit a office of normalised wet expanse for different pools having dissimilar initial masses (one thousand i ), drying at xx% humidity, T = 23 ± 1 °C, on the same surface, a tile.
Again, when normalised, to a first approximation, the curves overlap each other, corresponding to the fitting function: m/m i = 1 −α[1 − (A/A i )] β , with α = 0.78 and β = 0.16 This function gives an approximative relation betwixt the wet surface area and the mass of the pool. Hence, from A x , 1000 x is obtained. Finally t ten tin exist estimated using
$$\delta t=\frac{\delta m}{{J}^{\ast }{A}_{i}}$$
(12)
with δm =m i − thousand 10 . Given t i = 0 s, the expression of t x is:
$${t}_{x}=\frac{\blastoff R{yard}_{B}{T}^{ii}{A}_{i}^{\mathrm{1/2}}{h}^{\mathrm{1/2}}\rho {\mathrm{[1}-({A}_{x}/{A}_{i})]}^{\beta }}{Yard{d}^{two}\pi {P}^{\mathrm{1/ii}}{D}_{blood}{P}_{w}{P}_{a}}$$
(13)
A drying time of 8 h 18 min was calculated for the photo in Fig. 12, which was only 19 min less than the verbal time 8 h 37 min. The process was repeated for another puddle dried in the same conditions, and a difference of 5% was obtained.
By repeating this process several times as the pool dries, an average value would reduce the uncertainty. This first issue of calculation of the fourth dimension at which a pool was formed is a promising outcome, suggesting that it will become possible to calculate the time at which a pool was formed on a law-breaking scene. For at present, many precautions should be taken. This calculation worked for a controlled environment. Anyway, by further investigating the dissimilar variables, mainly the temperature which tin influence the evaporation rate, some reference values could be established to create a reference table. A ready has already been envisaged that would only require a camera. Pictures of the drying front of the puddle in different areas, depending on the accessibility, could exist automatically taken at a regular interval (such as every v or 10 min). This could exist done for a couple of hours and then later on prototype processing, a statistically valid value could be obtained with a margin of error. Therefore, if a reference tabular array existed for the dissimilar temperatures, humidity, and surfaces, a defined protocol could exist set. Although, work should be done in order to evaluate if analysing only some parts of the advancing front gives equivalent results as for entire blood pools. The method presents as well some limitations, since to work, the drying front must exist present. This ways, that if the pool is already fully dried, the method tin no longer be applied. On the contrary, if the puddle is yet entirely wet, then the investigation teams would have to wait for the drying front to appear. Thus, further work has now to be focused on the influence of the previously described parameters, and on the development of a very precise image processing tool. Indeed, since the difference between wet area and dried expanse is dependent on the paradigm analysis, hereafter development should seek to improve this tool.
Conclusions and perspectives
In this study the evaporation dynamics of blood have been described in particular and some similarities with the sol-gel transition were establish. Indeed claret is similar to a colloidal suspension with RBCs existence the dispersed stage, and plasma the aqueous stage. Afterward coagulation and fibrin atmospheric precipitation, blood forms a gel similar organization that volition and so dry following dissimilar evaporation rates. This evaporation rate has been compared to the evaporation charge per unit of pools of water drying in like conditions. Finally, some of the pool characteristics were assessed such as the size and the shape of the pool. A blood pool is an intermediate system betwixt a drop and a larger stretch of liquid. The latter corresponds to a system involving ane dimensional rut and mass transfers. For the blood puddle the evaporation at the triple line influences the unabridged evaporation rate. As a result we found that the shape influences the rate, which is expressed in a shape factor. The size of a pool, of class interferes with evaporation since, the greater is the volume that has to evaporate. Moreover, the drying front of the pool evolves in a non-uniform manner since it can start evolving towards the centre on one side while on the other side the pool is yet in a gel like phase. Ultimately, all these parameters are linked to the advancing drying front end. We have investigated the evolution of the drying front and related it to the evaporation rate of h2o, J *. By using the wet surface area of the pool to calculate J *, an nigh constant value of the evaporation rate was observed. Then by incorporating the different variables that act int the evaporation procedure of a pool, an approximative abiding value of improvidence coefficient was observed for pools of diverse sizes and shapes, but drying in the same weather. Using this approximation, it became possible to calculate the time at which a pool, dried in the aforementioned weather condition, was formed. Nonetheless, this last correlation should be investigated further for more pools, of different shapes and larger sizes, and drying at different temperatures and humidities. Some try on the research should be turned towards this issue as it could and then bring a modelling of the evaporation of blood pools, which could then be monitored on a crime scene simply by following the drying front end and using a reference table. This study used blood from good for you volunteers, males and females with haematocrit values ranging from 36.two to 47.1%. Private biological parameters did not testify any significant influence on the evaporation dynamics. However other physiological parameters such illnesses, like haemophilia, or the intake of drugs, similar aspirin, could have an indirect influence on evaporation since they are known to interfere with clotting. Hereafter piece of work on individual physiological aspects would thus better the proposed model. However, this investigation has demonstrated pioneering results apropos the drying of blood pools from good for you subjects, which may observe exploitation in forensic analysis.
The following work is going through a patent application at the European Patent Office. The inventors of this patent are F. Smith, C. Nicloux, D. Brutin. The awarding number is EP19305772.6. The patent was filed on the 17th of June 2019. The patent awarding covers the forensic interpretation section.
References
-
Brodbeck, S. Introduction to Bloodstain Pattern Analysis. J. Police Sci. Practice, 51–57 (2012).
-
Lee, H. C. Estimation of original book of bloodstains. Identification news xi–12 (1986).
-
Sant, S. P. & Fairgrieve, S. I. Exsanguinated Blood Book Estimation Using Fractal Assay of Digital Images: Claret Volume Estimation. J. Forensic Sci. 57, 610–617 (2012).
-
Laan, North. et al. Bloodstain Blueprint Analysis: implementation of a uid dynamic model for position determination of victims. Sci. Rep. five (2015).
-
Laan, N., Bremmer, R. H., Aalders, K. C. & de Bruin, One thousand. G. Book Conclusion of Fresh and Dried Bloodstains by Ways of Optical Coherence Tomography. J. Forensic Sci. 59, 34–41 (2014).
-
Bartz H. F. Estimating orginal bloodstain book: the development of a new technique relating volume and surface area, PhD thesis, Laurentian University, Sudbury, Ontario (2003).
-
Lund, Due south. P. & Lyer, H. Chiliad. Likelihood ratio as weight of forensic bear witness: a closer look. J. Res. Natl. Inst. Stan. 122, 27 (2017).
-
Laan, N., Smith, F., Nicloux, C. & Brutin, D. Morphology of drying claret pools. Forensic Sci. Int. 267, 104–109 (2016).
-
Bourges-Monnier, C. & Shanahan, K. E. R. Inuence of Evaporation on Contact Angle. Langmuir 11, 2820–2829 (1995).
-
Birdi, K. S., Vu, D. T. & Winter, A. A study of the evaporation rates of pocket-size water drops placed on a solid surface. J. Phys. Chem. 93(nine), 3702–3703 (1989).
-
Picknett, R. Yard. & Bexon, R. The evaporation of sessile or pendant drops in still air. J Colloid Interf. Sci. 61(2), 336–350 (1977).
-
Hu, H., Larson, R. G. & Magda, J. J, Measurement of wall-slip-layer rheology in shear-thickening wormy micelle solutions. J. Rheol. 46(4), 1001 (2002).
-
Brochart-Wyart, F. Droplets: Capillarity and Wetting. Soft Matter Phys. (pp. 15–eighteen. Springer, Berlin Heidelberg, 2013).
-
Johnston, B. M., Johnston, P. R., Corney, Due south. & Kilpatrick, D. Non-Newtonian blood ow in homo right coronary arteries: steady state simulations. J. Biomech. 37, 709–720 (2004).
-
Smith, F. R. & Brutin, D. Wetting and spreading of human blood: Contempo advances and applications. Curr. Opin. Colloid In. 36, 78–83 (2018).
-
Gijsen F. J. H. & Allanic Eastward., The inuence of the non-Newtonian properties of blood on the ow in large arteries: unsteady ow in a curved, J. Biomech. 10 (1999).
-
Kolbasov, A. et al. Blood rheology in shear and uniaxial elongation. Rheol. Acta 55, 901–908 (2016).
-
Brinker C. J. & Scherer K. W. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing. Elsevier Scientific discipline (1990).
-
Sherwood, T. One thousand. The Drying of Solids - I. Ind. Eng. Chem 21, 12–16 (1929).
-
Moore, F. The mechanism of moisture movement in clays with item reference to drying – a concise review. Brit. Ceram. Trans. J. 60, 517–539 (1961).
-
Macey, H. H. Clay-water relationships and the internal mechanism of drying. Brit. Ceram. Trans. J. 41, 73–121 (1942).
-
Dwivedi, R. Grand. Drying behaviour of alumina gels. J. Mat. Sci. lett. five(4), 373–376 (1986).
-
Wootton, D. K. & Ku, D. N. Fluid Mechanics of Vascular Systems, Diseases, and Thrombosis. Annu. Rev. Biomed. Eng. one, 299–329 (1999).
-
Sobac, B. & Brutin, D. Desiccation of a sessile drop of claret: Cracks, folds germination and delamination. Colloids Surface A 448, 34–44 (2014).
-
Pauchard, L., Parisse, F. & Allain, C. Inuence of salt content on fissure patterns formed through colloidal suspension desiccation. Phys. Rev. East 59(three), 3737 (1999).
-
Pauchard 50., Adda-Bedia K., Allain C., & Couder Y. Morphologies resulting from the directional propagation of fractures, Phys. Rev. E 67 (2003).
-
Lazarus V. & Pauchard L. From craquelures to spiral cleft patterns: inuence of layer thickness on the crack patterns induced by desiccation. Soft Matter seven, no, 2552 (2011).
-
Brutin D., Sobac B., Loquet B. & Sampol, J. Pattern formation in drying drops of blood, J. Fluid Mech. 667 (2011).
-
Triolet J. & Sallé B. Évaluation de la vitesse d'évaporation et de la concentration d'un composé organique volatil dans l'atmosphére d'un local de travail, Aide-mémoire technique INRS, 13 (2009).
-
Sobac B. & Brutin D. Structural and evaporative evolutions in desiccating sessile drops of blood. Phys. Rev. East 84 (2011).
-
Pauchard L. & Allain C. Stable and unstable surface evolution during the drying of a polymer solution drop. Phys. Rev. Eastward 68 (2003).
Author information
Affiliations
Contributions
F. Smith wrote the principal manuscript, realised the experiments, the presented graphs, and the suggested interpretation of the obtained results. C. Nicloux brought specialist knowledge and expertise on bloodstain pattern analysis. D. Brutin supervised the presented work. All authors reviewed the manuscript.
Respective author
Ideals declarations
Competing interests
The authors declare no competing interests.
Additional data
Publisher's note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed nether a Creative Commons Attribution 4.0 International License, which permits utilise, sharing, adaptation, distribution and reproduction in any medium or format, every bit long equally y'all give appropriate credit to the original writer(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other 3rd party cloth in this article are included in the commodity'southward Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons license and your intended utilise is not permitted past statutory regulation or exceeds the permitted use, you lot will need to obtain permission straight from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this commodity
Smith, F.R., Nicloux, C. & Brutin, D. A new forensic tool to date man claret pools. Sci Rep x, 8598 (2020). https://doi.org/10.1038/s41598-020-65465-4
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/s41598-020-65465-iv
Comments
By submitting a annotate you agree to abide by our Terms and Community Guidelines. If you lot notice something calumniating or that does not comply with our terms or guidelines please flag information technology equally inappropriate.
Source: https://www.nature.com/articles/s41598-020-65465-4
0 Response to "Rh- Dried Blood Add Solution and It Is Alive Again"
Post a Comment